 
Standard absolute entropy values are given in units of joules per kelvin per mole J K-1 mol-1 Values of standard absolute entropy (S) for many substances have been tabulated. We write \(S^o_A\left(T\right)\) to indicate the absolute entropy of substance \(A\) in its standard state at temperature \(T\). The standard absolute entropy of a substance, S, is the absolute entropy of a substance in its standard state (298.15 K, 100 kPa). It is usually included in compilations of thermodynamic data for chemical substances. I think youre right that entropy as a singular, absolute value is highest at high temperature, but the change in entropy is maximized the lower the initial. The standard molar entropy of a substance is the absolute entropy of 1 mole of the substance in the standard state. The standard entropy is usually given the symbol \(S^o\). As long as the change in the heat capacity as a function of temperature is similar between two substances, then the relative ranking based on absolute entropy will be similar to a ranking of the internal energy, which is what the author means when he says that the former can be used as 'a rough indicator or approximate index' of the latter. When the entropy value is calculated for one mole of the substance in its standard state, the resulting absolute entropy is called the standard entropy. Where the substance undergoes phase changes, the contribution that the phase change makes to the entropy of the substance is equal to the enthalpy change for the phase change divided by the temperature at which it occurs.Īt any given temperature, the entropy value that is obtained in this way is called the substance’s absolute entropy or its third-law entropy. 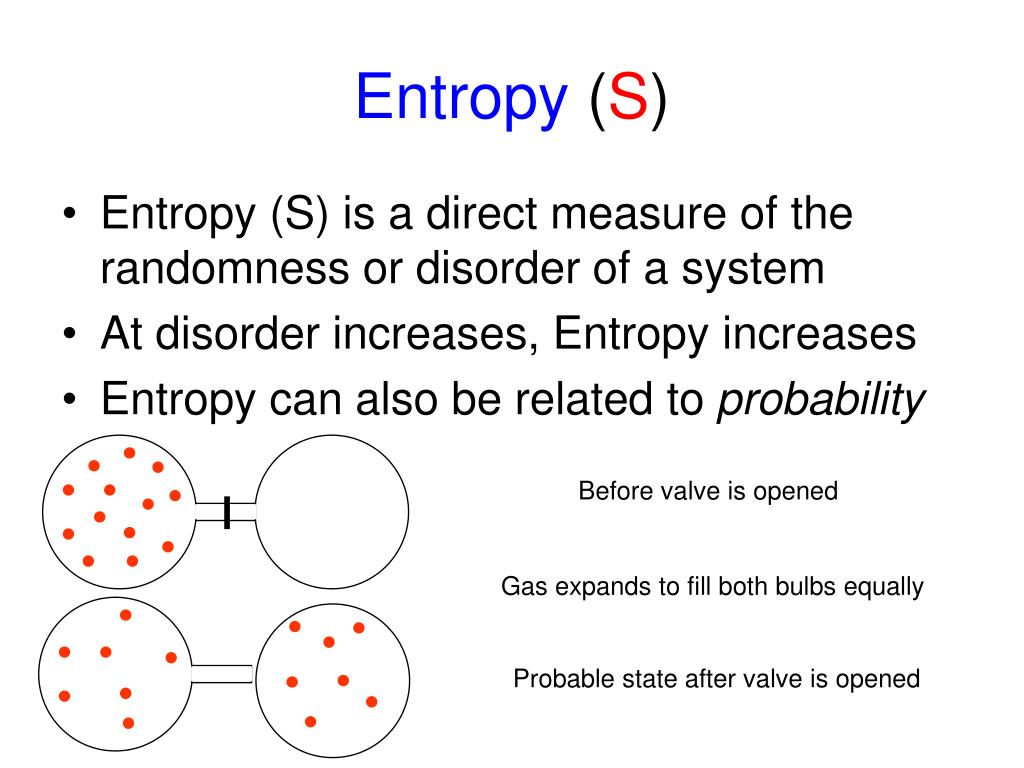
This video solution was recommended by our tutors as helpful for the problem above. In which of the following processes does entropy decrease for the system (a) boiling water to form steam (c) mixing of two gases into one container (b) dissolution of solid KCl in water (d) freering water to form ice 3. NH3( g) Ne( g) SO2( g) CH3CH2OH( g) He( g) Verified Solution. Which one of the following substances has the highest absolute entropy at 25C (a) Nib() (b) Neg) (c) H:01) (d) C(s) 2. Phase changes are isothermal and reversible. the entropy of a pure perfect crystal at 0 K is zero, therefore we have absolute entropies. Rank each set of substances in order of increasing standard molar entropy (S°). In temperature ranges where experimental heat capacity data are available, the entropy change is obtained by integration using these data. \), using Debye’s theoretical relationship, \(C_P=AT^3\) \(A\) is obtained from the value of \(C_P\) at the lowest temperature for which an experimental value of \(C_P\) is available. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
